TU Munich
New semiconductor hybrid material splits water more efficiently
Until now, the low efficiency has slowed down the large-scale industrial use of electrolysis of water to produce sustainable fuels. An international team of researchers has now developed a catalyst for water splitting that is four times more efficient.
A research team from the Technical University Munich (TUM) has developed an efficient water splitting catalyst as part of an international cooperation. It consists of a double helix semiconductor structure coated with carbon nitride. This catalyst is ideal for cheap and sustainable hydrogen production. An important step towards climate neutrality.
The international team around the TUM chemist Tom Nilges and the engineer Karthik Shankar from the University of Alberta has succeeded in finding a stable yet flexible semiconductor structure that splits water much more efficiently than previously possible. The core of the structure is an inorganic double helix compound consisting of the elements tin, iodine and phosphorus (SnIP). It is synthesized in a simple process at temperatures of around 400°C. The compound is then processed in an inorganic double helix. SnIP fibers are on the one hand flexible and at the same time as robust as steel.
»The material combines the mechanical properties of a polymer with the potential of a semiconductor«, explains Tom Nilges, Professor of Synthesis and Characterization of Innovative Materials at the Technical University Munich. »This will enable us to manufacture flexible semiconductor components in a further technical step«.
Catalyst splits water efficiently

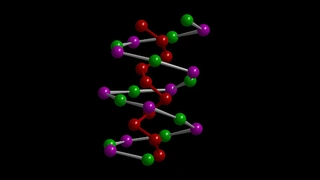
Soft carbon nitride shell, hard SnIP core
With the water splitting catalyst, the research team developed a first application for this unusual material. They produced nanoparticles from both starting substances and mixed the suspensions of these two nanoparticles. The result is a structure consisting of a hard yet flexible core of SnIP double helices encased in a soft shell of carbon nitride.
As measurements have shown, the resulting heterogeneous structure is not only much more stable than the starting materials, it can also split water four times more efficiently than before — and is so interesting as a material with which hydrogen can be cheaply produced or excess electricity from wind turbines can be chemically stored.
One-dimensional fibers increase surface area
The high efficiency of the catalyst is mainly due to its larger surface area. The team succeeded in increasing the surface area by dividing the SnIP fibers into finer strands. The most effective mixture is 30 percent SnIP with 70 percent carbon nitride.
The thinnest fibers consist of a few double helix strands and are only a few nanometers thick. The material is therefore practically one-dimensional. Wrapped in carbon nitride, it retains its high reactivity, but is more durable and therefore better suited as a catalyst.
Also interesting for optoelectronics
The one-dimensional SnIP double helices also open up completely different applications. It would be particularly exciting for researchers to have only one double helix strand of SnIP. This would then be available with clockwise or counterclockwise rotation — each with very special optical properties. This makes SnIP interesting for optoelectronics.
»We were able to show theoretically that many other compounds of this kind can exist and are currently working on the synthesis of these materials«, adds Nilges. »Flexible inorganic, nanometer-sized 1D semiconductors can cause as much hype as 2D layer materials such as graphene, phosphorus or molybdenum disulfide currently do.«





